Frequency
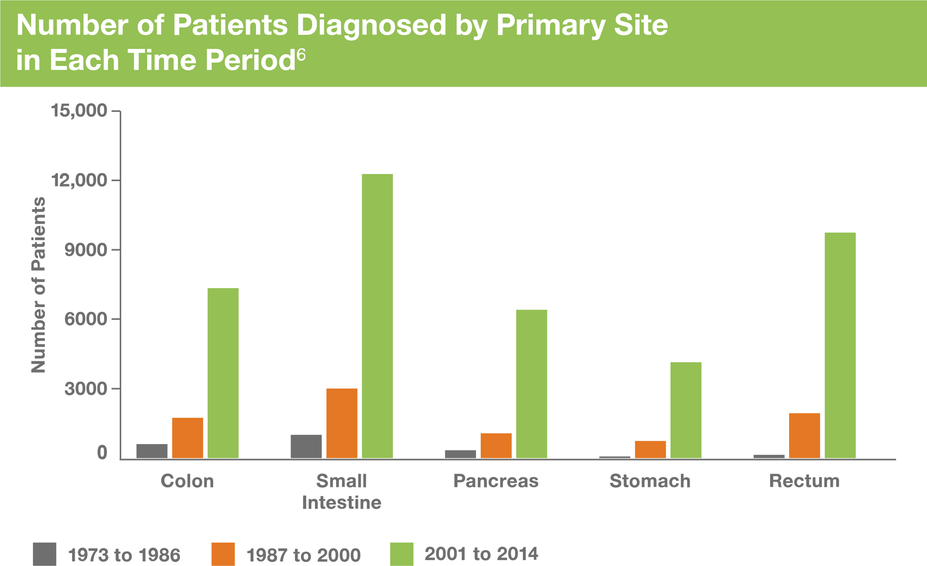
NETs Are Being Diagnosed With Increasing Frequency
Neuroendocrine tumors (NETs) arise from neuroendocrine cells, which are specialized cells that secrete hormones and other substances and are found throughout the body. These cancers may or may not secrete substances at levels high enough to cause symptoms.1-3
Although classified as a rare disease, NETs are being diagnosed with increasing frequency.4,5
The reported incidence of NETs has increased:
6.98/100,000 | New cases/year as of 20124 |
~171,000 | Projected people living with the disease in the United States as of 20144 |
~6.4-fold increase | Over the last few decades |
Diagnosis at earlier stages of the disease is thought to be responsible for this increase, even though NETs are still often diagnosed at an advanced or metastatic stage.7,8
NETs are generally considered as indolent (slowly growing) tumors; however, the prognosis varies widely, according to4:
Primary tumor site
Tumor differentiation and grade
Stage
From 1973 through 2012, the annual age-adjusted incidence of NETs has increased compared with that of other malignant neoplasms.4
Classification
Although NETs can arise from a wide variety of organs and tissues, most NETs are gastroenteropancreatic NETs (GEP-NETs), originating in the gastrointestinal tract and pancreas.4
GEP-NETs have traditionally been divided into10:
Foregut tumors: Esophagus, stomach, proximal duodenum, liver, and pancreas
Midgut tumors: Distal duodenum, ileum, jejunum, ascending colon, and proximal two-thirds of transverse colon
Hindgut tumors: Distal one-third of transverse colon, descending colon, sigmoid colon, and rectum
Delay in Diagnosis
Many patients with GEP-NETs are diagnosed with metastatic disease, which may result in increased disease burden and worse survival outcomes.4,8,11
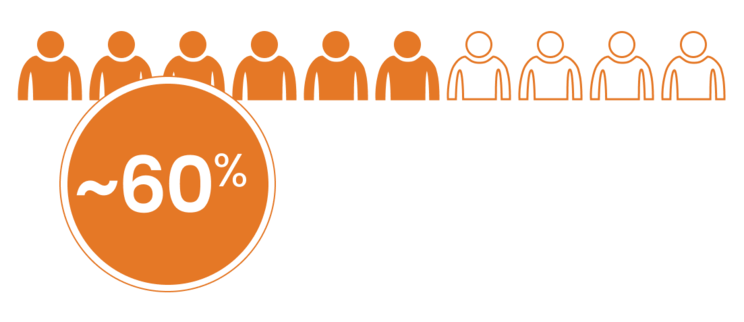
of patients with GEP-NETs present with regional spread or distant metastases at time of diagnosis8,a
aBased on 837 assessable patients from the National Cancer Registry for Gastroenteropancreatic Neuroendocrine Tumors (RGETNE) in Spain. In this cohort, 14% of patients had regional spread and 44% had distant metastases.8
Mortality
Advanced GEP-NETs May Be Fatal, Even for Patients With Low-Grade Disease4
Most patients are diagnosed with locally advanced or metastatic disease due to the slow-growing nature of the tumor, lack of symptoms, or nonspecific symptoms11,12
GEP-NETs may be fatal in patients with advanced, nonresectable disease4,7